Myriad Neuroscience announced a new post hoc analysis of the GUIDED study focusing on the impact of the GeneSight® test on clinical outcomes for patients aged 65 years or older who took psychotropic medications.
The GUIDED (Genomics Used to Improve DEpression Decisions) study was the largest prospective study to assess the benefit of pharmacogenomics to inform treatment for depression using the GeneSight Psychotropic test versus a treatment as usual control arm who also received active therapy.
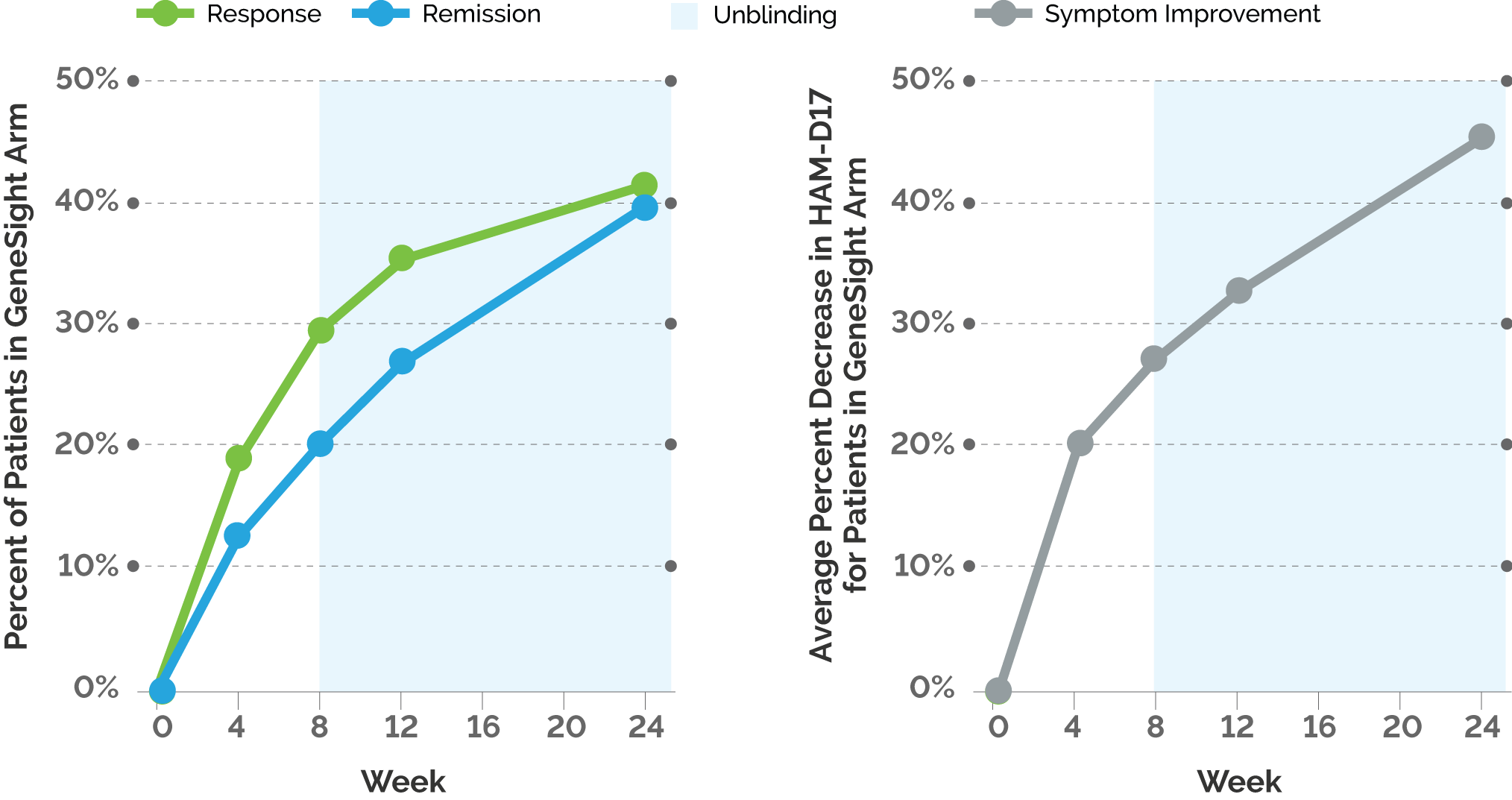
The study is a rater- and patient-blinded randomized controlled trial that evaluated outpatients diagnosed with major depressive disorder (MDD) and with a patient- or clinician-reported inadequate response to at least one psychotropic medication. The GUIDED study was an 8-week, blinded, randomized controlled trial of 1,167 subjects from 20 academic sites and 40 community sites. The trial was unblinded after the 8-week check-in, and the subjects in the GeneSight arm were followed out to 24 weeks.
Why Depression Treatment is Important for Older Adults
According to the Centers for Disease Control (CDC), an estimated 7 million of the nation’s 39 million adults aged 65 years and older are affected by depression, which can be described as a “persistent sad, anxious, or empty feeling, or a feeling of hopelessness and pessimism” that lasts for longer than two weeks.
Geriatric depression puts substantial burdens on function, quality of life, and healthcare resources. A study published in the American Journal of Geriatric Psychiatry found that depression among older adults is linked with longer length of illness, more depressive episodes, and a greater number of comorbidities.
Additionally, late-life depression is often accompanied by cognitive impairment, dementia, and chronic medical conditions. A broad range of clinical and social factors complicate its presentation and medical management.
When treating this population, clinicians may face many challenges, such as increased polypharmacy, lower adherence, and higher rates of side effects.
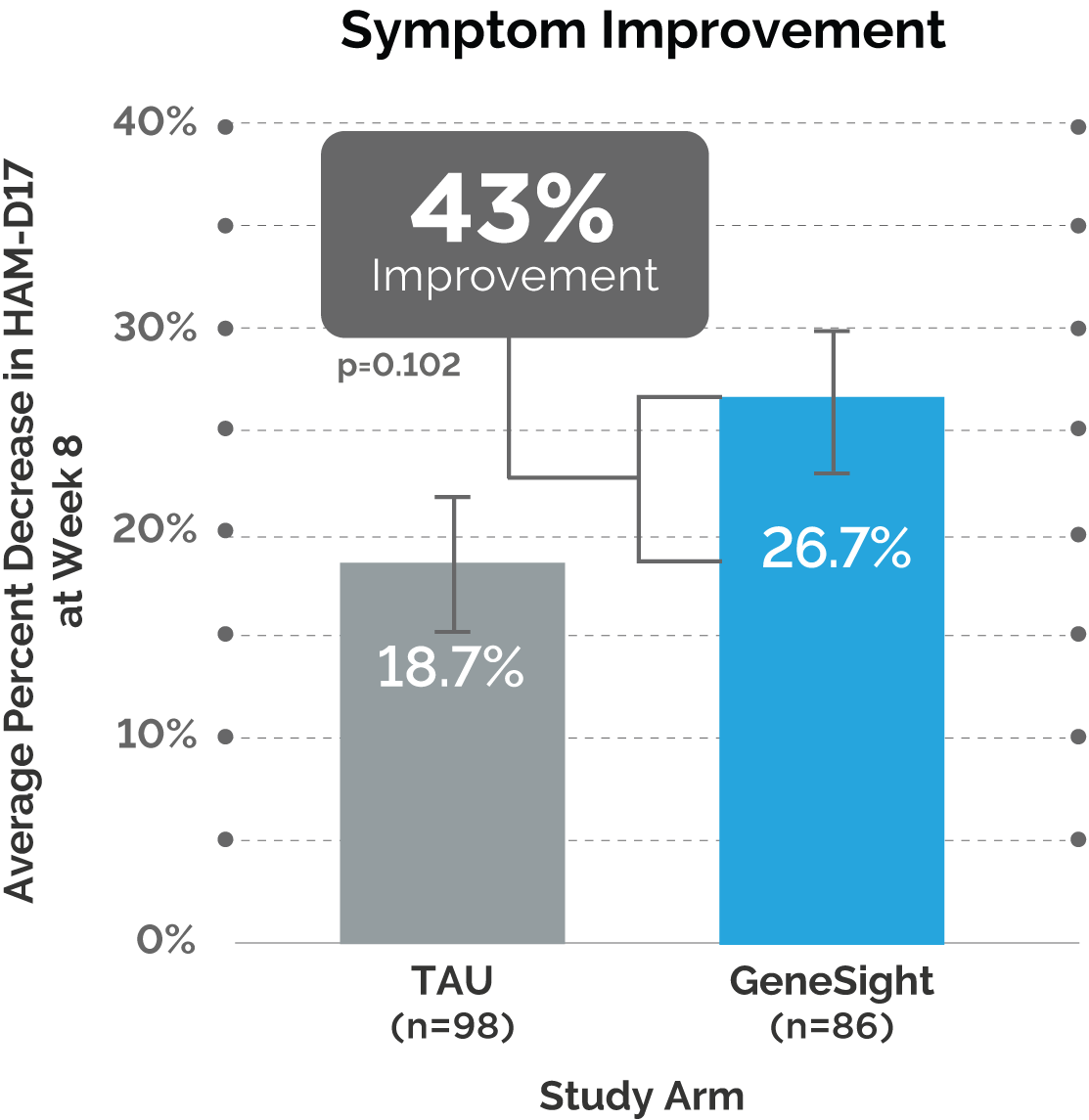
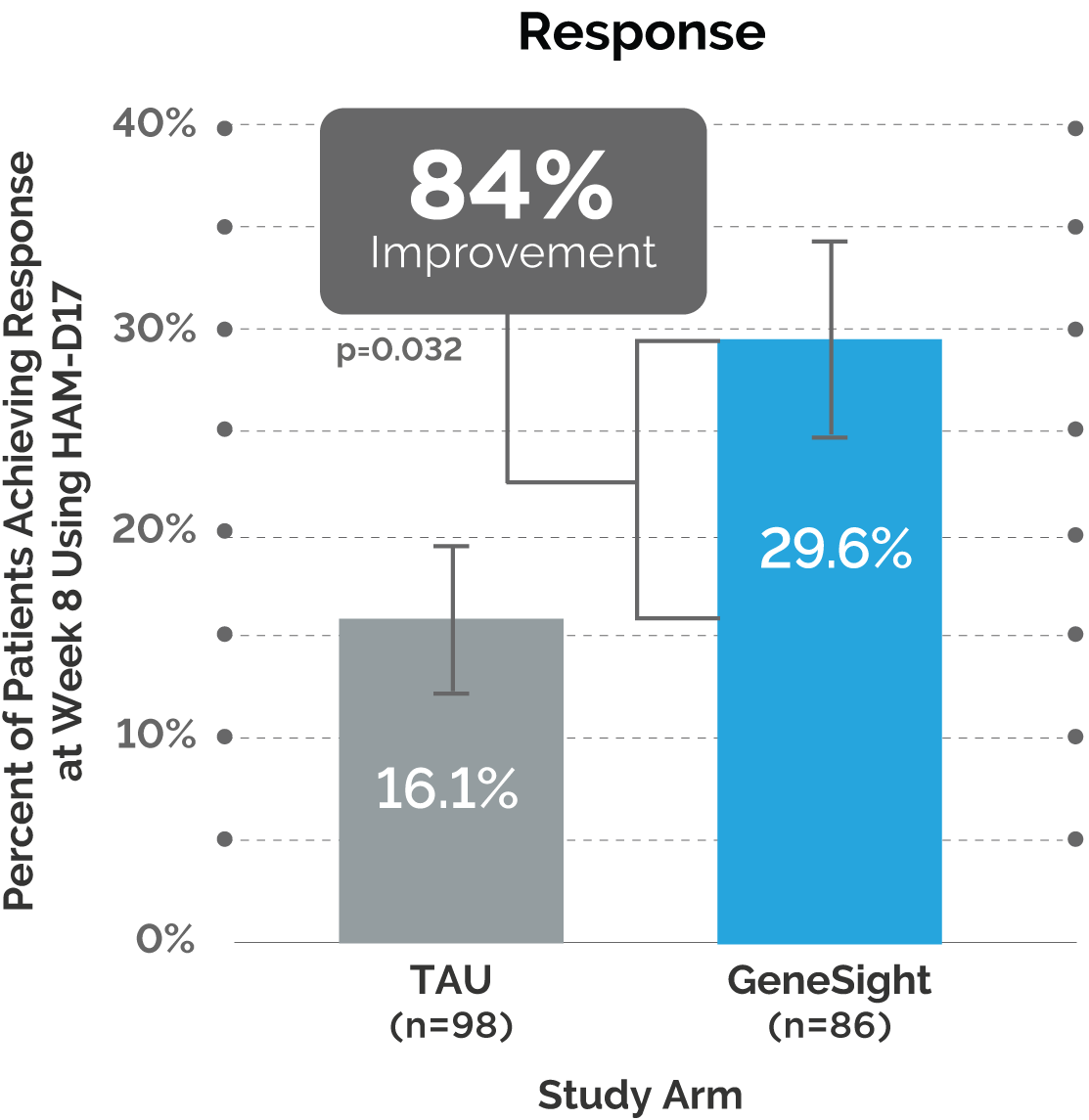
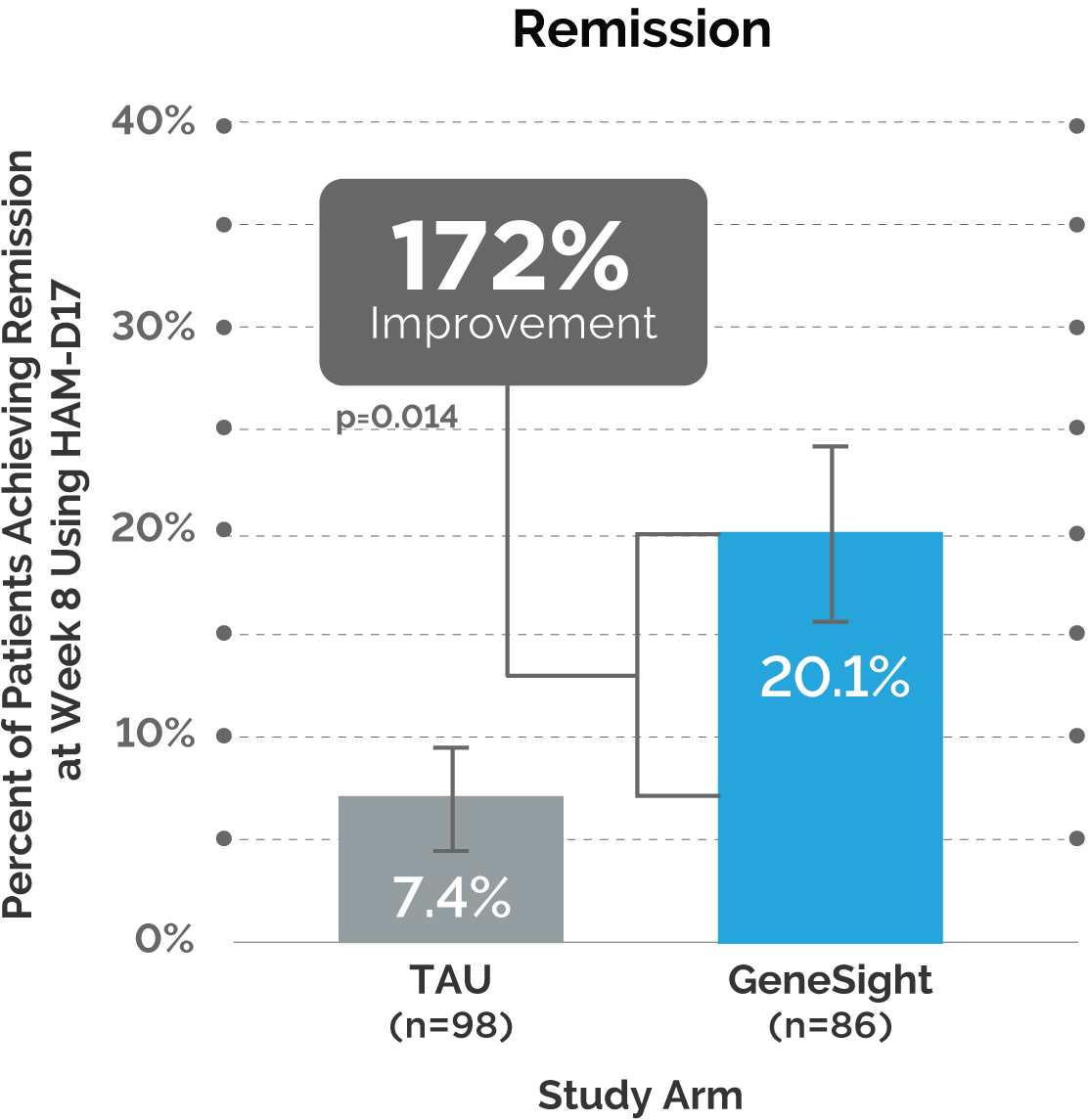
Results of the GUIDED 65+ Analysis
Next, the post hoc analysis showed outcomes continued to improve over time for patients in the GeneSight® arm who were 65 years or older.
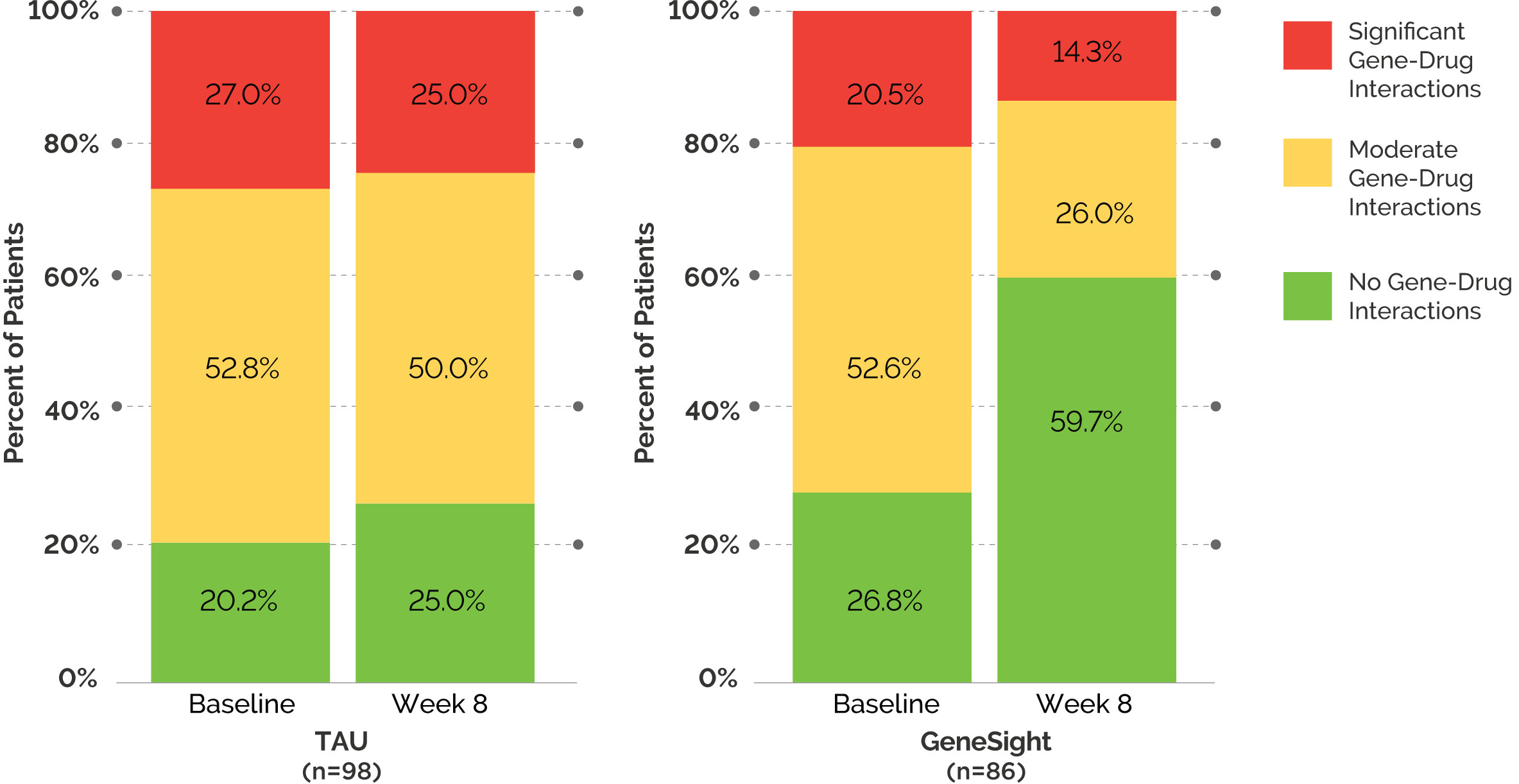
Finally, a larger proportion of patients in the GeneSight arm were taking medications with no gene-drug interactions by week 8 compared to TAU.
It’s important to note that not all patients who receive the GeneSight Psychotropic test will see improvements in symptoms, achieve response, or achieve remission.
Depression May Present Differently in Older Adults
This post hoc analysis of the GUIDED study is important because how depression presents in geriatric-aged patients, as well as how it is treated, may be different than in younger patients.
For example, elderly patients may have impaired production of CYP450 enzymes, which can affect drug metabolism rate. This may exacerbate the effect of pharmacogenomic variation. The poor metabolizer phenotype may be more extreme in elderly patients compared to younger patients. For example, one study showed that serum levels of venlafaxine among elderly CYP2D6 poor metabolizers were 8-fold greater than younger CYP2D6 poor metabolizers.